KPV: COSPEP's Ultimate Anti-Inflammatory Peptide
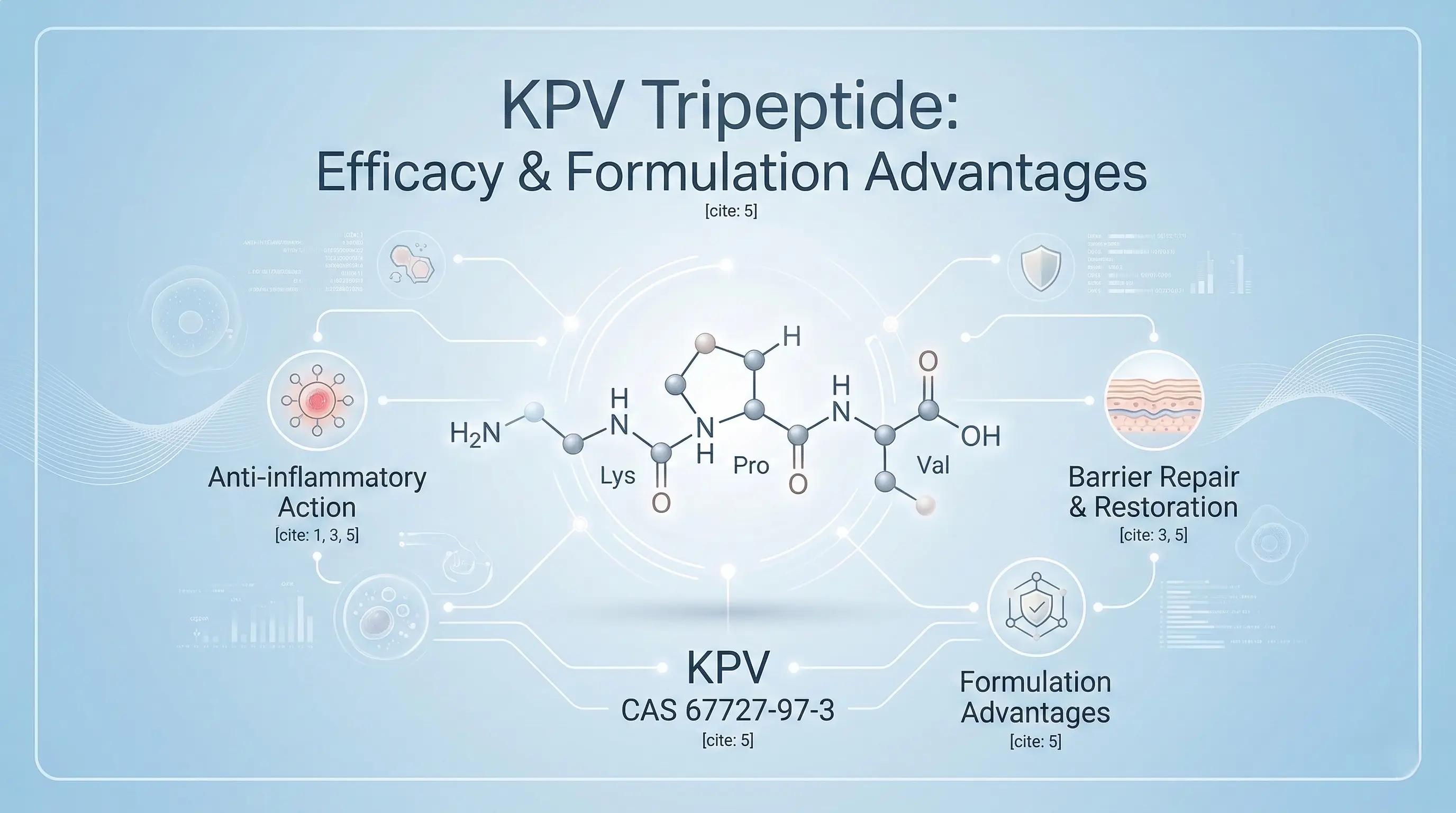
The KPV tripeptide (CAS 67727-97-3), derived from α-MSH via structural optimization, is a premier cosmetic active ingredient. It dissociates anti-inflammatory efficacy from melanogenesis, inhibits the NF-κB pathway intracellularly, and has superior formulation advantages (342 Da MW, targeted adhesion, stability, amphiphilicity). COSPEP supplies high-purity KPV for advanced skincare.
COSPEP Technical Overview
The skincare and cosmetic dermatology sectors consistently seek therapeutic agents capable of delivering significant anti-inflammatory efficacy without concurrent adverse effects. While traditional soothing agents often present limitations in efficacy, and long-term use of clinical-grade anti-inflammatories may compromise epidermal barrier integrity, endogenous peptides offer a promising alternative. Among these, the KPV tripeptide (CAS 67727-97-3) has emerged as a premier active ingredient.
Derived from the structural sequence of a naturally occurring human hormone, KPV functions as a potent immunomodulatory agent. Its primary distinguishing characteristic is the complete dissociation of anti-inflammatory action from melanogenesis (skin darkening)—an outcome that historically limited the application of earlier melanocortin-based technologies. This report outlines the molecular engineering of KPV and its distinct advantages in modern cosmetic formulation.
1. Biological Origins: The Dual Functionality of α-MSH
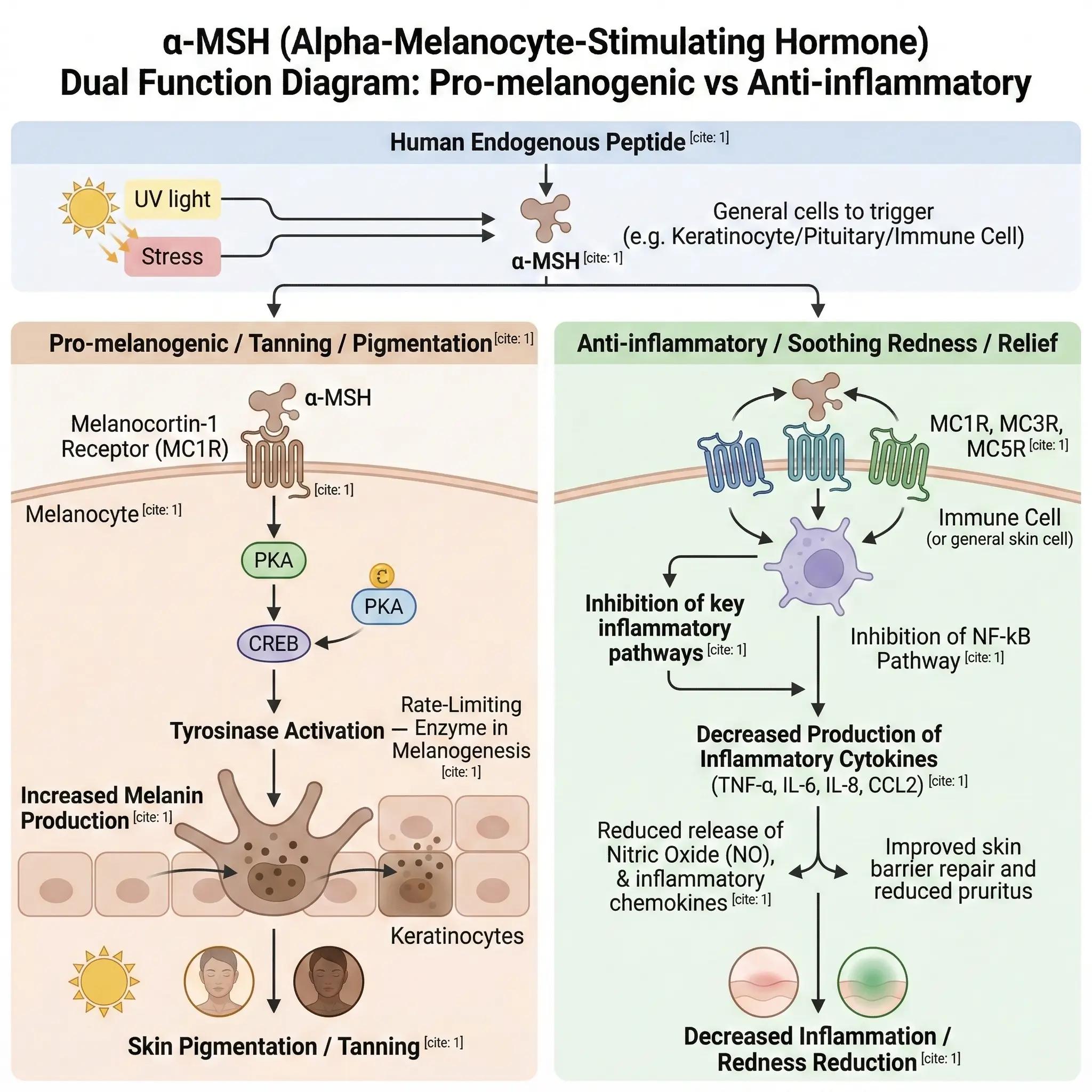
The developmental rationale for KPV originates from the biological mechanisms of α-melanocyte-stimulating hormone (α-MSH). α-MSH is an endogenous, 13-amino-acid peptide secreted by the human body in response to physiological stressors, such as ultraviolet (UV) radiation or chemical trauma.
Dual function diagram of α-MSH
Historically recognized primarily for its role in stimulating melanogenesis (the tanning response) to protect cellular DNA, subsequent scientific research elucidated a secondary, concurrent function. α-MSH acts as one of the body's most potent endogenous anti-inflammatory mediators, capable of rapidly attenuating localized immune hyperactivity and modulating the release of pro-inflammatory cytokines.
2. Clinical and Commercial Limitations of α-MSH
The initial identification of α-MSH's anti-inflammatory properties presented significant potential for dermatological applications, ranging from post-procedure erythema reduction to the management of atopic dermatitis. However, a critical physiological limitation impeded its clinical and commercial viability: the dosage required to achieve therapeutic anti-inflammatory effects inherently triggered the hormone's primary melanogenic pathway, resulting in visible hyperpigmentation.
From a dermatological and consumer standpoint, inducing skin darkening to mitigate inflammation is generally contraindicated. The industry required a methodological approach to isolate the peptide's immunomodulatory properties from its melanogenic activity.
3. Structural Optimization: Precision Peptide Cleavage
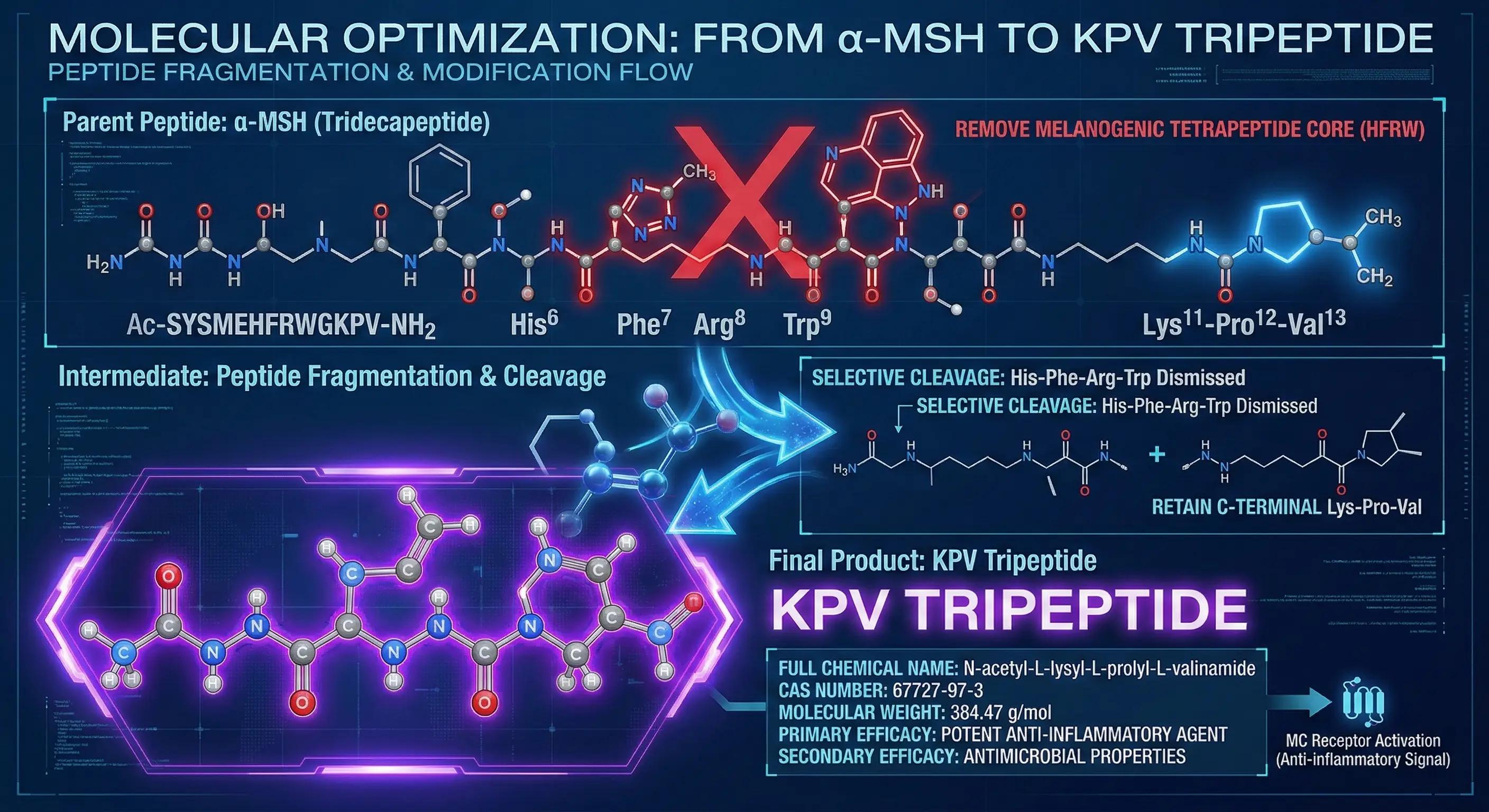
To address this limitation, researchers comprehensively analyzed the structure-activity relationship (SAR) of the α-MSH molecule. The central tetrapeptide sequence was identified as the primary pharmacophore responsible for activating the melanocortin-1 receptor (MC1R) and initiating the melanin synthesis cascade.
By selectively excluding this central region and isolating only the three amino acids at the C-terminus—Lysine, Proline, and Valine (KPV)—biochemists successfully engineered a truncated analogue. Empirical testing demonstrated that this refined tripeptide retains the profound anti-inflammatory and barrier-repairing capabilities of the parent molecule. Most significantly, due to the omission of the requisite receptor-binding sequence, KPV does not induce melanin production, thereby successfully dissociating therapeutic healing from undesirable hyperpigmentation.
4. Formulation Advantages and Physicochemical Properties
Beyond its biological efficacy, the KPV tripeptide possesses physicochemical characteristics that render it highly advantageous for cosmetic and pharmaceutical manufacturing. It is optimized to address common formulation challenges:
- Ultra-Low Molecular Weight for Transdermal Penetration: With a molecular weight of approximately 342 Da, KPV exhibits optimal transdermal penetrability. This intrinsic ability to bypass the stratum corneum reduces the dependency on aggressive chemical penetration enhancers, allowing the active agent to reach viable epidermal and dermal layers efficiently.
- Targeted Adhesion via Lysine: The N-terminal Lysine residue carries a positive charge at physiological pH. Given that inflamed or damaged cellular microenvironments frequently exhibit an increased negative surface charge, this characteristic facilitates targeted electrostatic adhesion to compromised tissues.
- Enhanced Stability via Proline: Peptide degradation is a pervasive challenge in formulation science. The central Proline residue features a rigid pyrrolidine ring, providing a structural conformation that significantly increases resistance to proteolytic enzymes. This structural rigidity translates to improved shelf-life stability and a prolonged biological half-life upon topical application.
- Amphiphilic Profile via Valine: The lipophilic nature of the C-terminal Valine, juxtaposed with the hydrophilic Lysine, confers an amphiphilic profile to the KPV molecule. This dual solubility profile facilitates seamless incorporation into diverse formulation matrices, including aqueous serums, multi-phase emulsions, and lipid-rich creams.
5. Mechanism of Action: Intracellular NF-κB Inhibition
The pharmacological mechanism of KPV provides a robust scientific foundation for its anti-inflammatory efficacy. Due to its diminutive size and specific peptide transporters, KPV is capable of intracellular translocation, where it exerts its primary effect by inhibiting the Nuclear Factor kappa B (NF-κB) pathway.
NF-κB functions as a primary transcription factor mediating the inflammatory cascade. Upon exposure to environmental stressors, chemical exfoliants, or light-based dermatological therapies, NF-κB activation induces the synthesis of pro-inflammatory cytokines (such as IL-1β, IL-6, and TNF-α), resulting in erythema and edema. KPV effectively stabilizes the inhibitory complex associated with NF-κB, suppressing this cascade at the transcriptional level. This mechanism provides rapid, targeted mitigation of inflammatory symptoms without engaging the melanogenic pathways associated with cell-surface receptor binding.
6. Conclusion and Strategic Implementation
The development of the KPV tripeptide represents a significant advancement in biomolecular engineering and dermatological science. By offering a stable, highly efficacious, and safe alternative to traditional anti-inflammatory agents, KPV is uniquely positioned to serve as a cornerstone active ingredient in advanced skincare formulations.
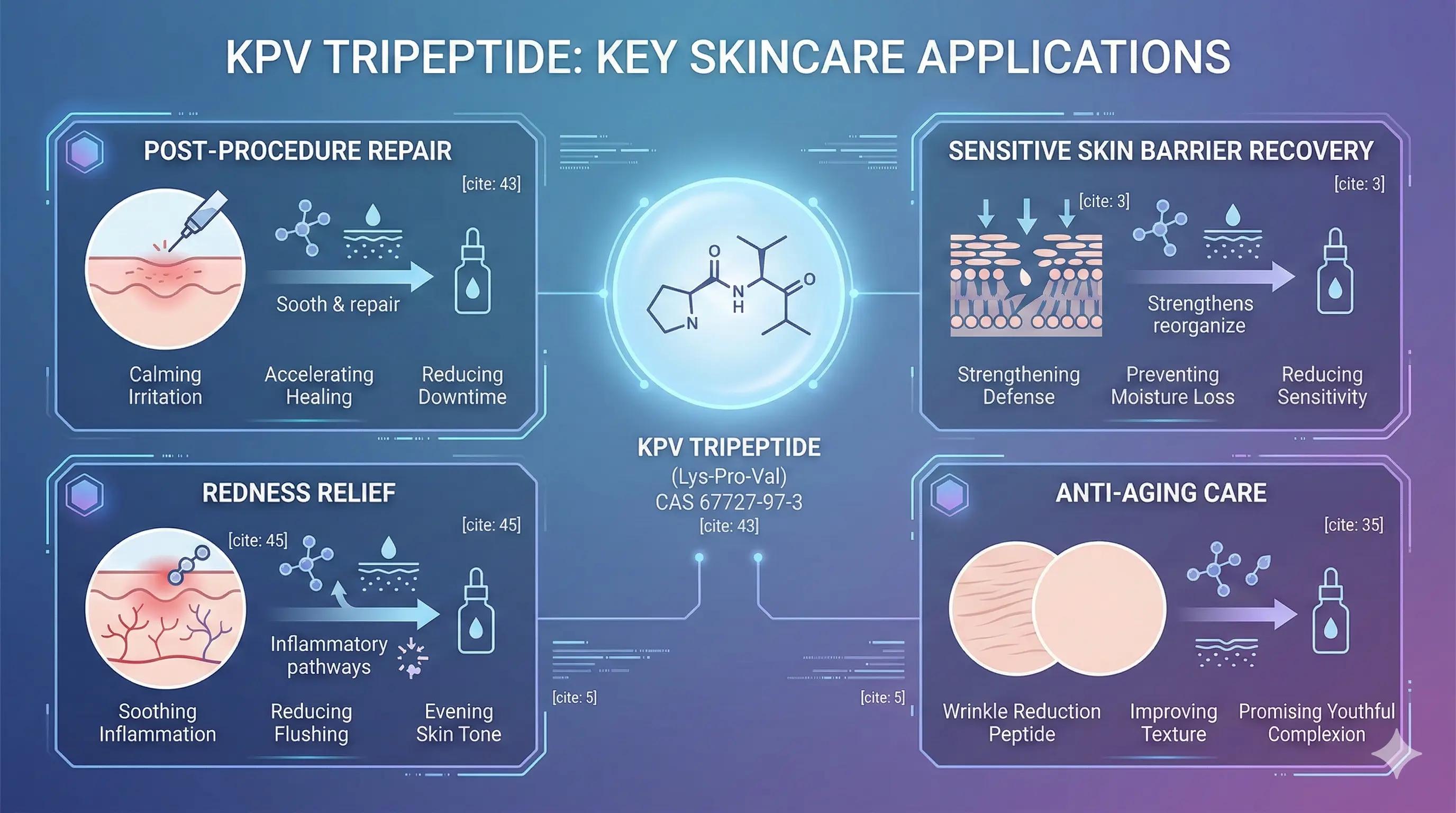
Whether integrated into post-procedure recovery serums, daily barrier-repair treatments for sensitive skin, or advanced anti-aging regimens, KPV provides a tangible technological and therapeutic advantage. COSPEP supplies high-purity KPV synthesized to meet rigorous formulation standards, facilitating the development of next-generation dermatological and cosmetic solutions.