
Practical Procurement Guide for B2B Buyers: How to Choose Between Peptide Vials and Bulk Lyophilized Powder
Learn how professional buyers evaluate peptide vials versus bulk lyophilized peptide powder for industrial formulation, manufacturing scalability, purity correction, and cost efficiency.
In the peptide and cosmetic active ingredient industry, many buyers assume that a "5mg vial" and a "1g peptide raw material" are essentially the same product in different packaging. While chemically they may originate from the same upstream peptide synthesis batch, commercially and industrially, they are fundamentally different products.
For B2B buyers—including cosmetic formulators, R&D laboratories, compounding facilities, contract manufacturers, and raw material distributors—understanding this distinction is critical for controlling formulation accuracy, production scalability, actual active dosage, cost efficiency, and regulatory risk.
1. The Two Product Types Are Built for Different Purposes
A. Peptide Vials (5mg / 10mg / 20mg)
A vial product is usually designed as a finished dosage-style presentation rather than a manufacturing raw material.
| Feature | Description |
|---|---|
| Packaging | Sterile vial |
| Quantity | Milligram-level |
| Form | Lyophilized cake/powder |
| Intended use | End-use or laboratory reconstitution |
| Common buyers | Small labs, clinics, resellers |
| Added excipients | Often yes |
| Unit cost | High |
| Scalability | Poor |
⚠️ Crucial Note: Many vial products include mannitol, trehalose, acetate buffers, stabilizers, or bulking agents. Therefore, a "5mg peptide vial" does NOT necessarily mean the total powder weight inside the vial is 5mg. The total lyophilized cake may weigh 20-50mg, while the actual peptide content is only 5mg.
B. Bulk Lyophilized Peptide Powder (1g / 5g / 10g+)
Bulk peptide powder is closer to an industrial active ingredient or API-like intermediate.
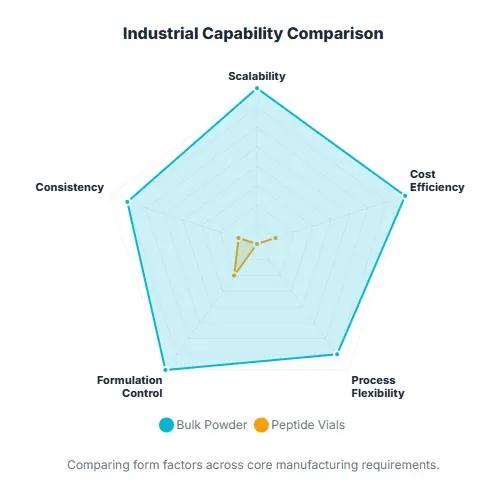
cospep Industrial Capability Comparison
| Feature | Description |
|---|---|
| Packaging | Glass bottle or foil bag |
| Quantity | Gram-level or higher |
| Intended use | Manufacturing and formulation |
| Common buyers | Cosmetic manufacturers, R&D teams |
| Concentration | High |
| Scalability | Excellent |
| Cost efficiency | Much better |
| Process flexibility | High |
For B2B procurement, unless the buyer specifically requires pre-filled vials, sterile presentation, or single-use convenience, bulk raw material is generally the correct choice.
2. The Biggest Industry Misunderstanding: “99% Purity”
Many buyers incorrectly assume: 99% Purity = 99% Active Content.
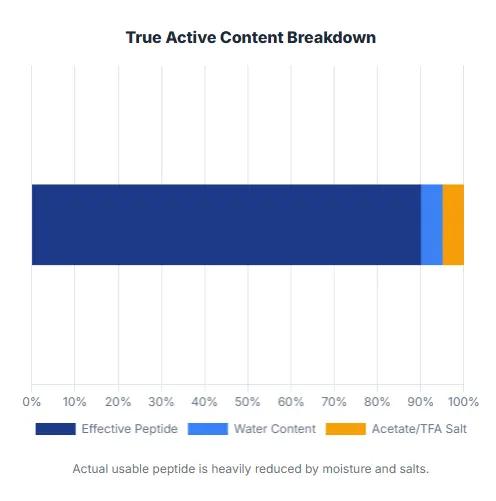
peptide powder purity vs acitve content
In reality, peptide COAs (Certificates of Analysis) often report HPLC Purity, which is not the same as the true Net Peptide Content.
A peptide raw material's real data might look like this:
| Parameter | Value |
|---|---|
| HPLC Purity | 99.0% |
| Water Content | 8.0% |
| Acetate Content | 5.0% |
| Net Peptide Content | 86% - 90% |
This means the actual usable peptide may be significantly lower than the advertised HPLC purity. For industrial formulation, recognizing this distinction is extremely important.
3. Core Logic and Formulas for Industrial Formulation
Most formulation errors occur because buyers calculate dosage using HPLC purity alone. The correct industrial workflow is:

The Industrial peptide Formulation Workflow
Effective Purity Formula
P_effective = P_assay x (1 - moisture) x conversionFactor
(Effective Purity = HPLC Purity × (1 - Moisture) × Salt Correction Factor)
Industrial Formulation Weight Formula

Peptide Content Equation
4. Real Manufacturing Example: Cosmetic Peptide Formulation
Suppose a formulator wants to manufacture 100kg (100,000g) of serum containing 2000ppm of a peptide. The peptide raw material parameters are:
- HPLC Purity: 99%
- Moisture: 5%
- Salt Correction: 96%
Step 1: Calculate Effective Peptide Content (P_effective)
0.99 x 0.95 x 0.96 = 0.9029 (or 90.29%)
Step 2: Convert ppm and calculate the required pure active peptide
2000ppm = 0.002
100,000g x 0.002 = 200g
(The formula actually requires 200g of pure active peptide)
Step 3: Calculate Required Raw Material (W_raw)
W_raw = 200g / 0.9029 = 221.51g
Conclusion: The manufacturer must accurately weigh 221.51g of the peptide raw material to achieve the target active concentration in the final serum batch.
5. Why Vials Are Usually a Poor Choice for Manufacturing
Many new buyers consider purchasing multiple peptide vials instead of bulk powder because the vial appears more "finished", more "sterile", or seems to have higher purity. However, for industrial production, vial products create several problems:
| Issue with Vials | Impact on Industrial Production |
|---|---|
| Inconsistent fill weights | Dosage variability and instability |
| Added excipients | Unknown formulation interference |
| High packaging cost | Poor economics, higher production costs |
| Small-scale packaging | Manufacturing inefficiency |
| Difficult batch scaling | Severe manufacturing limitations |
6. What Professional Buyers Should Actually Request
Instead of only asking, "What is the purity?"
Professional buyers should request to review the following parameters:
| Test Item | Why It Matters |
|---|---|
| HPLC Purity | To understand the impurity profile |
| Net Peptide Content | The baseline for calculating actual usable peptide |
| Water Content | Hygroscopic correction |
| Acetate/TFA Content | Salt correction |
| Residual Solvents | Process cleanliness |
| Endotoxin | Biological contamination indicator |
| Sterility | For injection-related applications |
| COA + Chromatogram | Verification of authenticity |
7. Procurement Recommendations Summary
✅ Choose Peptide Vials If:
- You need ultra-small quantities.
- You require convenience packaging.
- You are performing preliminary laboratory/small-scale testing.
- You need pre-measured units.
✅ Choose Bulk Lyophilized Powder If:
- You are manufacturing cosmetic products.
- You require scalable production.
- You need accurate formulation and concentration control.
- You want lower production costs.
- You need industrial dosage consistency.
Final Industry Insight In peptide procurement, the most expensive mistake is not buying "low purity" material. The biggest mistake is misunderstanding what the purity number actually means. A peptide labeled "99% Purity" does not necessarily contain 99% usable peptide content. Professional peptide procurement requires effective purity correction, moisture adjustment, salt correction, and industrial formulation calculations. That is the difference between purchasing a peptide product and sourcing a true manufacturing raw material.