
The Ultimate 2026 Manufacturing Guide: The Industrial Truths and Applications of Blue Copper Peptide (GHK-Cu)
Drawing on our deep industrial expertise, this definitive guide strips away marketing hype to reveal the precise chemistry, quality tiers, and formulation protocols that distinguish true, high-efficacy GHK-Cu from cheap imitations.
Abstract
This comprehensive guide delves into the industrial reality of Blue Copper Peptide (GHK-Cu), demystifying the significant price disparities in the raw material market. By analyzing the molecular distinction between GHK and the chelated GHK-Cu complex, we establish why "High Quality equals High Cost" is a physical certainty rooted in Solid-Phase Peptide Synthesis (SPPS) and precise Chelation technology.
The article categorizes current market offerings into four distinct quality tiers—from "Imposter" mixtures to "Crystal Grade" purity—and exposes the risks of free copper ions in inferior products. Furthermore, it provides technical guidance for formulators on interpreting HPLC data beyond the single peak, calculating Net Peptide Content (NPC), and managing critical stability factors such as pH windows (5.5–7.0) and ingredient incompatibilities. Concluding with a 2026 outlook, this piece serves as an essential manual for decision-makers to distinguish between concept dusting and true clinical efficacy.
Keywords:GHK-Cu, Copper Tripeptide-1, Blue Copper Peptide, Copper Peptide Complex, Solid-Phase Peptide Synthesis (SPPS), Chelation Technology, Net Peptide Content (NPC), HPLC Purity, Stoichiometry, Skin Remodeling, Collagen Synthesis, Wound Healing, Anti-aging, Tissue Regeneration,Low Free Copper, Crystal Grade, Lyophilized Powder
Editor's Note:
In the skincare raw material market, the price disparity for GHK-Cu (Blue Copper Peptide) is immense. Prices range from "high cost-performance" options at a few thousand dollars per kilogram to pharmaceutical-grade crystals costing hundreds of thousands. This massive price gap often leaves brand owners and procurement teams confused.
As a raw material supplier with a deep background in manufacturing, we know the reality: In the world of GHK-Cu, high quality inevitably means high marginal costs. This article strips away marketing jargon to reveal the true industrial landscape of Blue Copper Peptide—from molecular structure and manufacturing processes to formulation applications.
1. A Manufacturing Perspective: What Exactly is GHK-Cu?
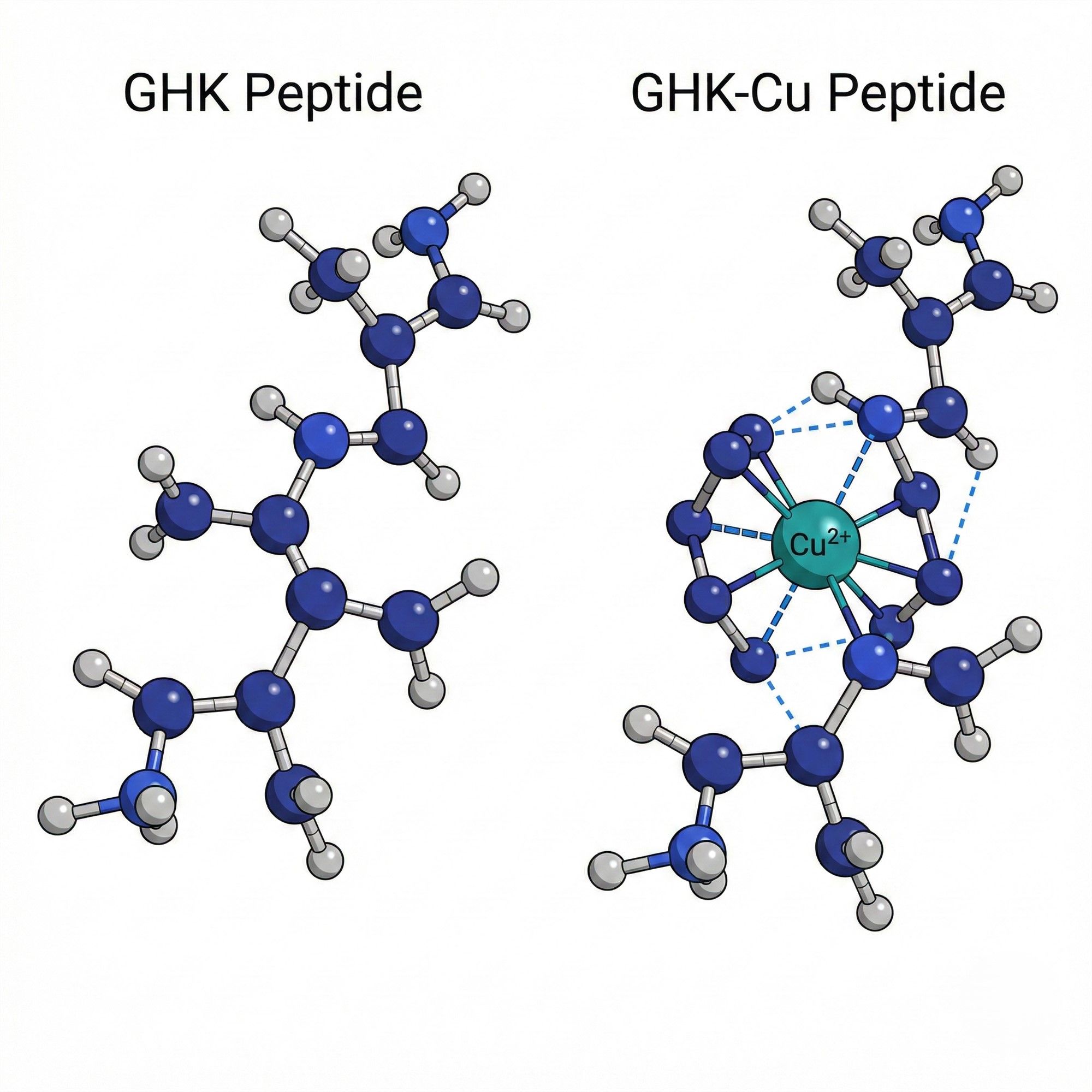
When we discuss Blue Copper Peptide (GHK-Cu), focusing solely on efficacy tags like "anti-aging" or "repair" often leads to overlooking the true technical core: the integrity of the molecular structure and the ability to control this structure during manufacturing.
1.1 Structural Differences: GHK vs. GHK-Cu
The primary task is to clarify the fundamental difference between GHK and GHK-Cu:
- GHK (Glycyl-L-Histidyl-L-Lysine): Essentially, this is a signal peptide sequence acting as an "empty" transport vehicle. Before binding with metal ions, its actual role in skin repair is relatively limited.
- GHK-Cu: The biologically active form is the stable complex formed by Chelation between GHK and divalent copper ions (Cu²⁺).
1.2 The Core Bottom Line: Containing Copper ≠ Copper Peptide
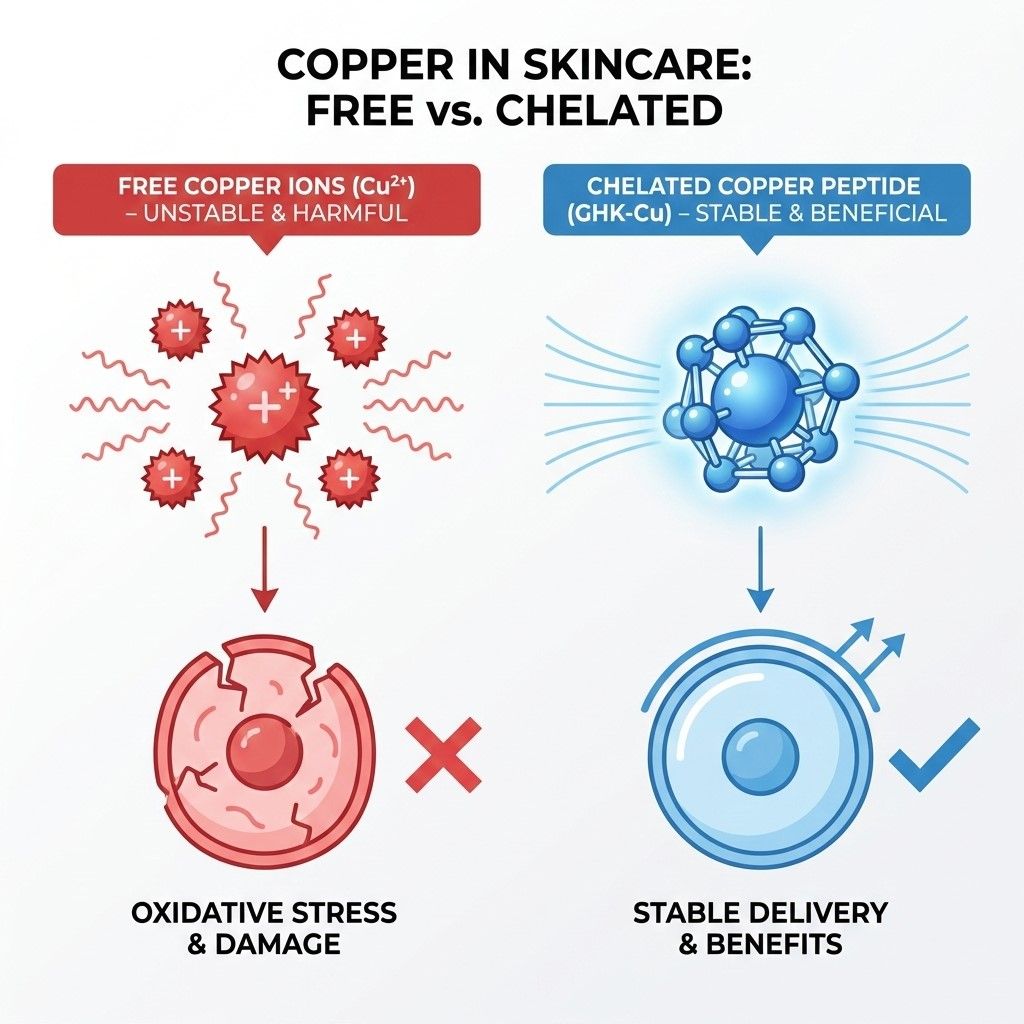
This transformation is not a simple physical mixture. It requires specific atoms on the peptide chain (Nitrogen/Oxygen) to form coordinate bonds that firmly "lock" the copper ion into a ring structure.
If a raw material simply mixes copper salts with peptides without forming this stable structure, the system will contain free copper ions. These free ions not only fail to repair the skin but also pose significant risks of Pro-oxidant activity and irritation.
Critical Warning: Color is Not a Quality Indicator
The blue color of a GHK-Cu solution is merely a byproduct of d-d electron transitions, not a proof of quality. The depth of the color is easily influenced by pH values or even artificially added dyes. True quality control lies in the extreme control of Stoichiometry—ensuring every single GHK molecule precisely captures one copper ion.
2. Industrial Manufacturing: Why High Quality Necessitates High Cost
The stratified pricing of GHK-Cu is not market hype but an inevitable result of the manufacturing path. The finer the control over the microscopic structure, the higher the marginal cost.
2.1 Synthesis Route: The Cost Logic of SPPS
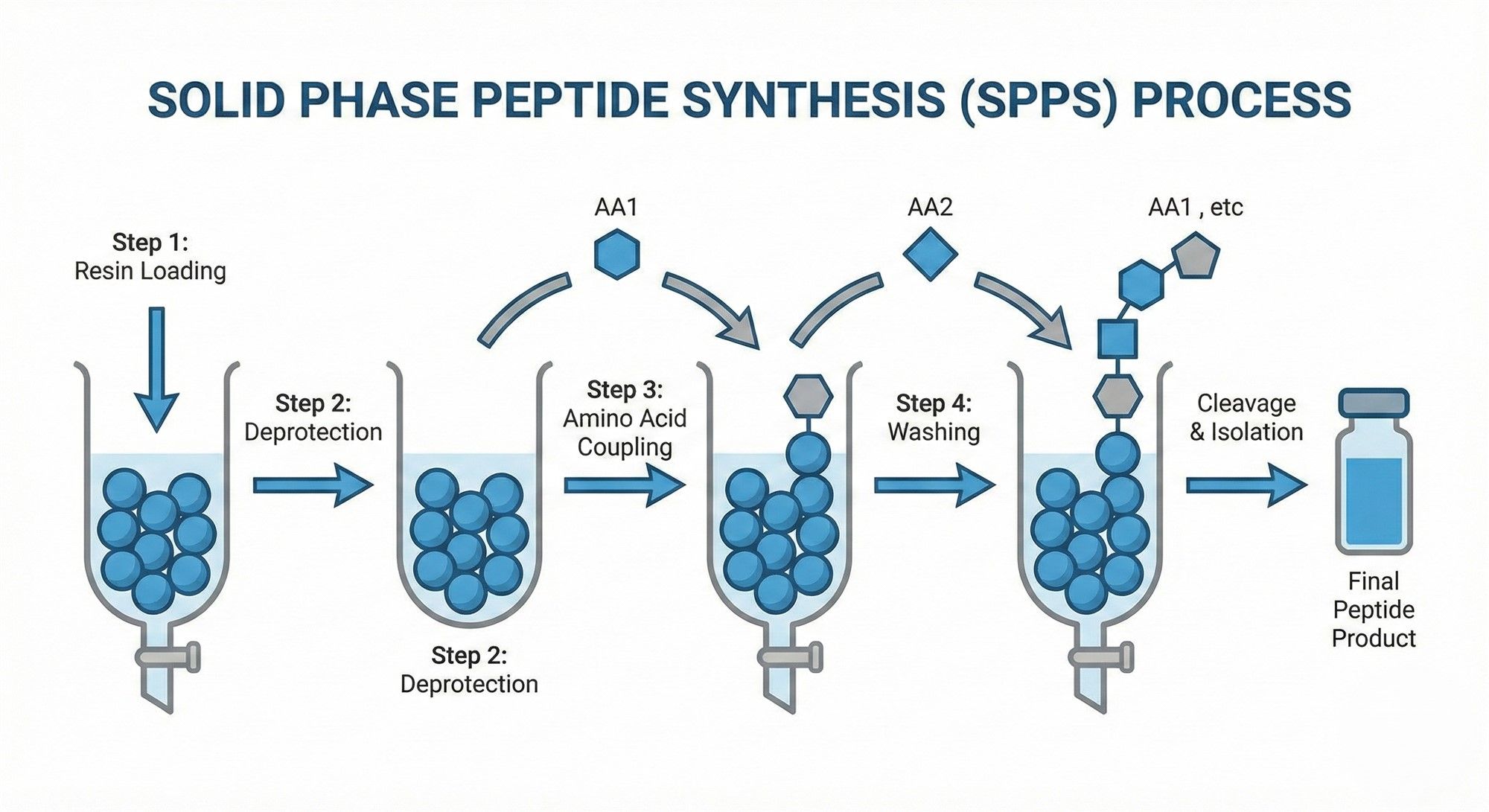
The mainstream method for producing high-quality GHK is Solid-Phase Peptide Synthesis (SPPS). While more expensive than liquid-phase synthesis, it allows for precise, block-by-block sequence control. The high costs stem from two main control points:
- Racemization Control: Preventing L-form amino acids from converting into inactive D-form impurities requires expensive catalytic systems.
- Deletion Sequence Control: To avoid short-chain byproducts, manufacturers must often use excess raw materials to drive the reaction to completion, sacrificing atomic utilization.
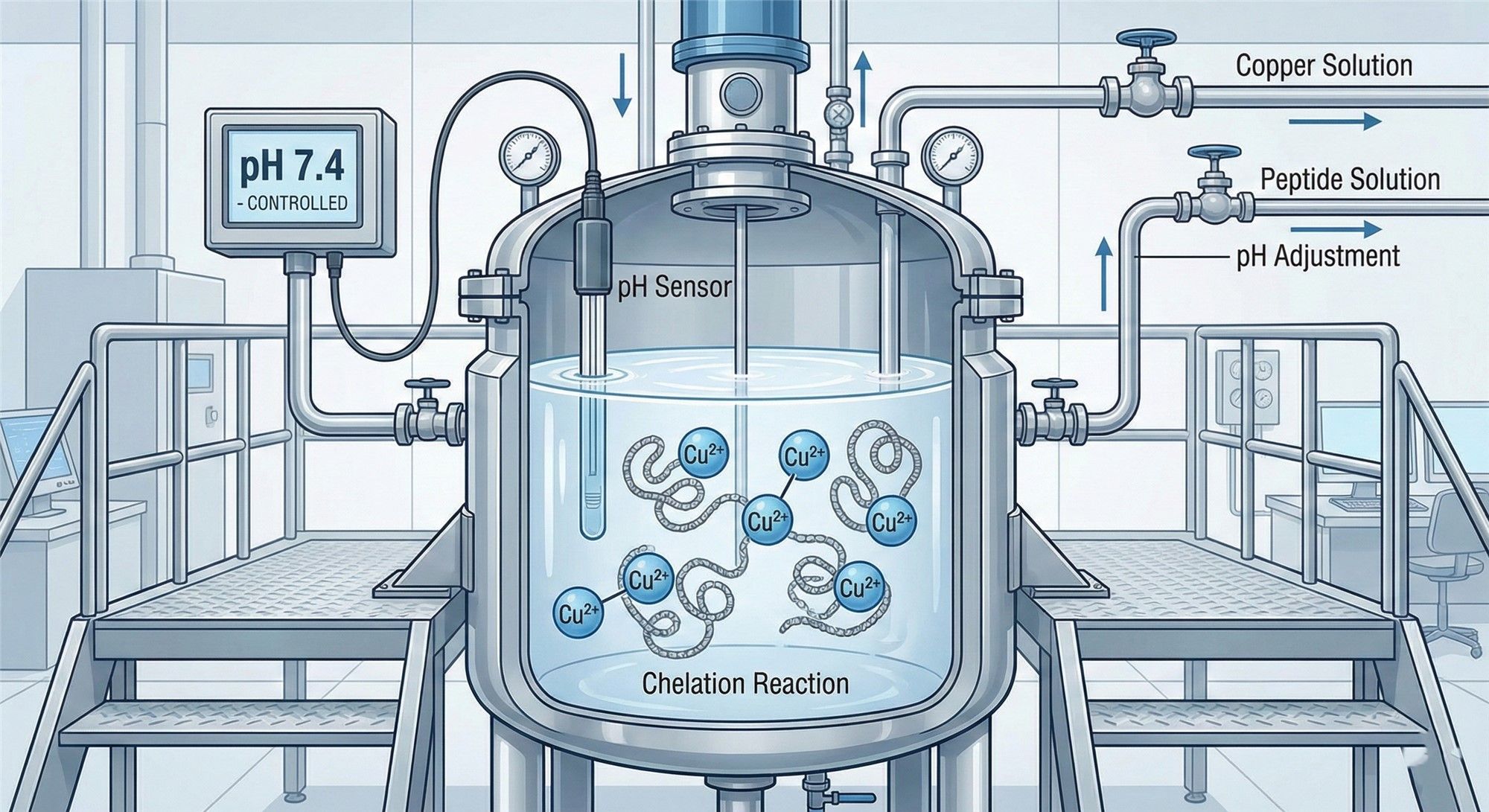
2.2 Chelation Process: The True Technical Watershed
After obtaining the peptide chain, the Chelation step determines the final activity. Real chelation processes have strict barriers:
- Copper Source Purity: Strict control over heavy metal impurities.
- Molar Ratio (1:1): Excess copper causes irritation; excess peptide leads to insufficient activity.
- Dynamic pH Control: Requires online monitoring systems to adjust protons released during the reaction in real-time, maintaining the specific pH window for coordinate bond stability.
2.3 Purification and Drying: The Last Mile of Stability
- RP-HPLC Purification: Manufacturers must "cut the head and tail," actively discarding fractions that contain the product but are impure. This represents a significant cost consumption.
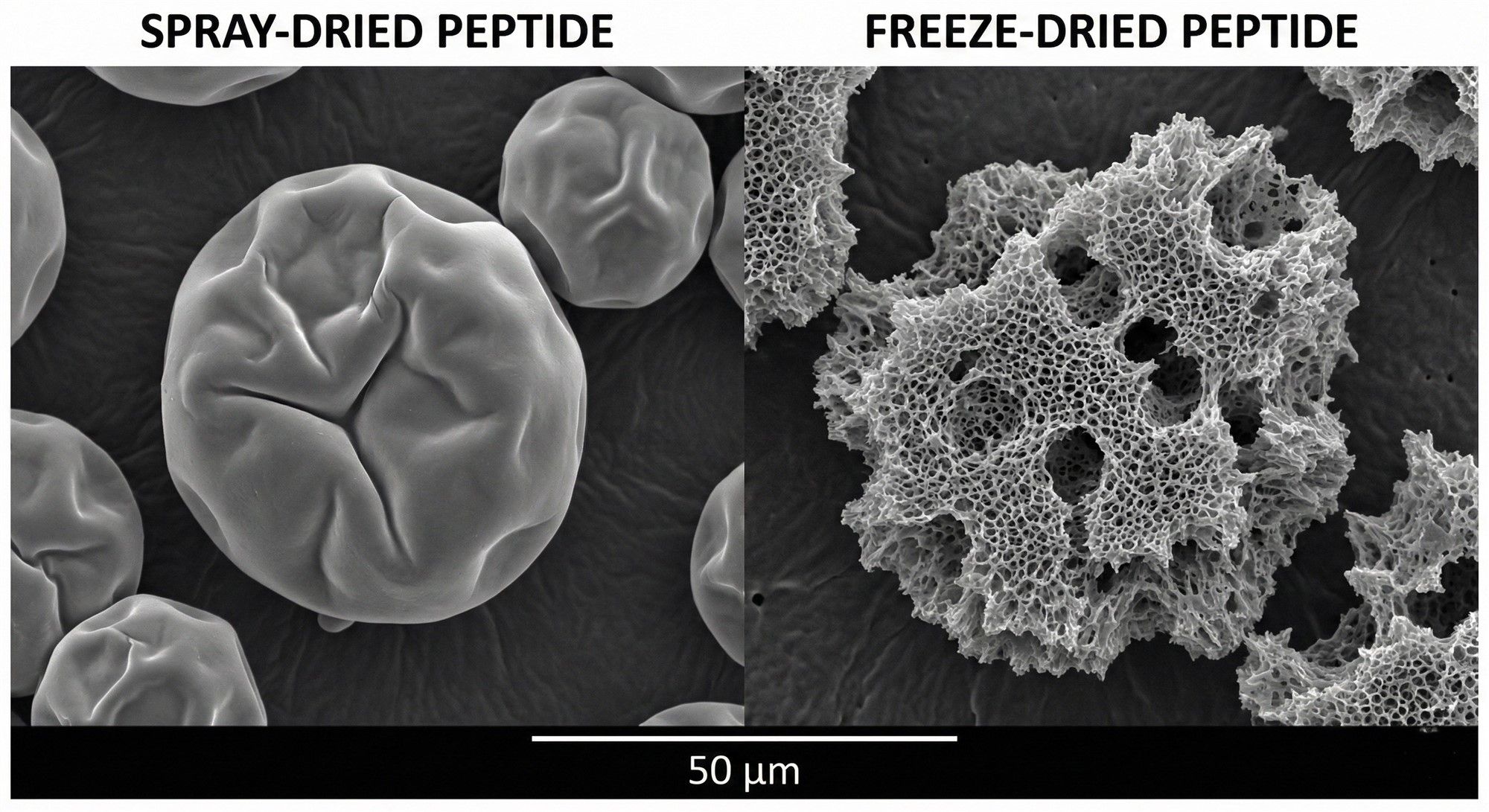
Drying Process: Cheap raw materials often use Spray Drying (high temperatures destroy activity); high-end materials use Lyophilization (Freeze-Drying). Although energy-intensive and slow, it preserves the loose, porous crystal structure and biological activity.
3. Market Guide: Which Grade of GHK-Cu Are You Buying?

To help procurement and technical personnel decode the COA (Certificate of Analysis), we categorize GHK-Cu on the market into four real tiers:
| Quality Tier | Typical Features | Essence & Risk |
|---|---|---|
| Tier 1: The Imposter | Extremely low price; color is unusually vivid and consistent. | Essence: Cheap short peptides + Copper salts + Dye. |
| (Concept Grade) | / | Risk: A complete waste of money. Ineffective and potentially carries heavy metal risks. |
| Tier 2: The Crude Mixture | Low price; powder is dull or greenish; pH is acidic. | Essence: Physical mixture skipping chelation and purification steps. |
| (Physical Mix) | / | Risk: High levels of free copper. Pro-oxidant tendency; causes sensitization and skin irritation. |
| Tier 3: Standard Grade | Medium price; RP-HPLC purity 95-98%. | Essence: Proper chelation but average purification precision. |
| (Precipitation Grade) | / | Application: Cost-sensitive mass-market products (dosage <0.1%). Long-term stability is limited. |
| Tier 4: Crystal Grade | Expensive; loose porous crystals; purity >99%. | Essence: Multiple purifications, strict 1:1 chelation, removal of counter-ions. |
| (Ultra-Pure) | / | Value: Extreme structural stability and batch consistency. The choice for high-end formulations. |
4. HPLC and "Invisible Quality Risks"
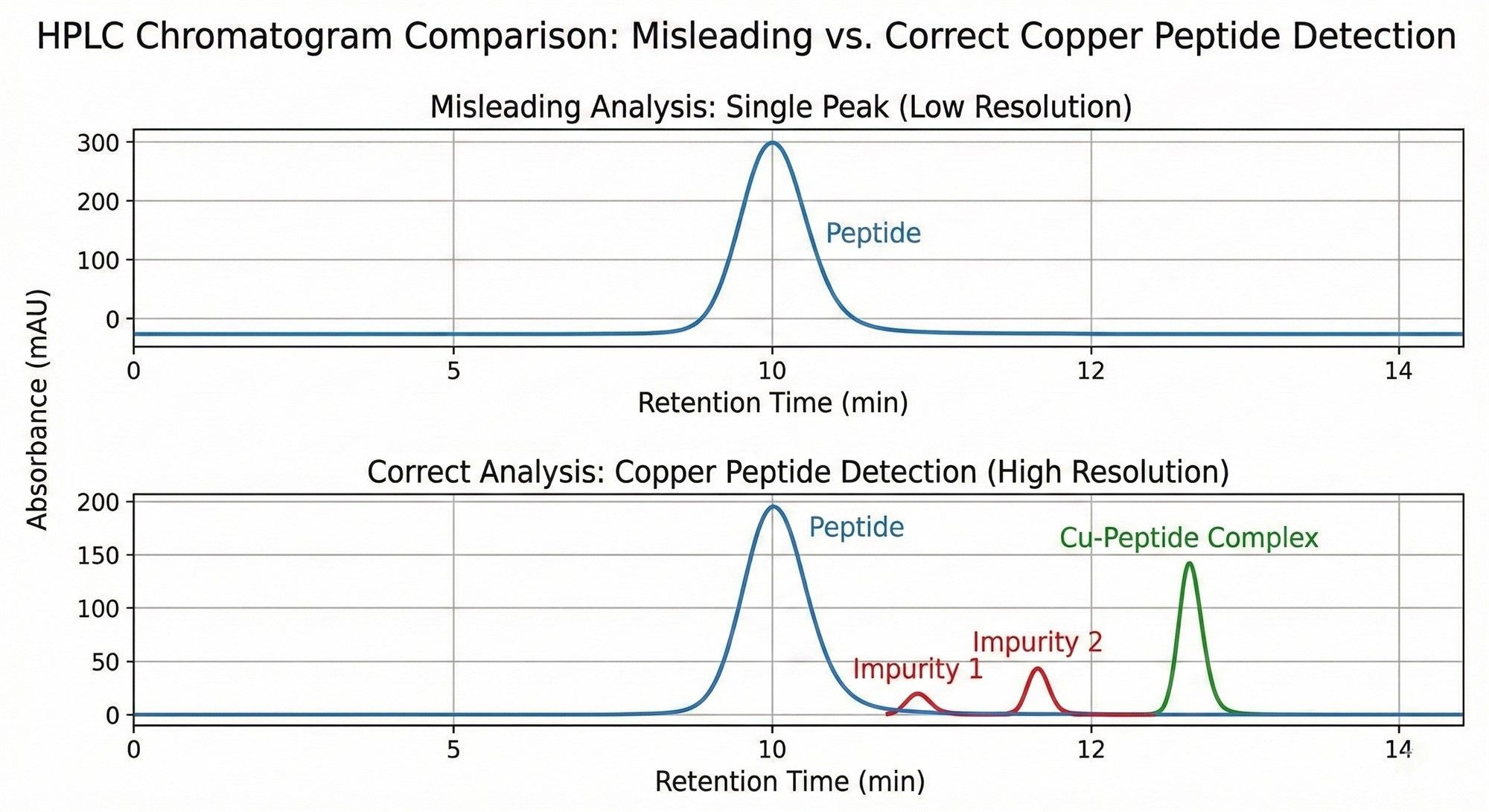
In quality control, do not be misled by a single towering peak. The HPLC chromatogram may be hiding a "numbers game."
- The Misconception of the Single Peak: If the detection method is improper (e.g., the mobile phase is too acidic), GHK-Cu will dissociate. The 99% peak you see might just be the GHK naked peptide, while the copper ions have been washed away.
- The Truth About NPC (Net Peptide Content): Purity $\neq$ Content (Assay). 1g of powder includes counter-ions (acetate) and bound water.
- Inferior Raw Material: NPC is only 60%-70%.
- Premium Raw Material: NPC is controlled at 80%-85%.
- Consequence: For the same 0.1% formula dosage, using inferior raw materials effectively reduces the active input by 30%. Furthermore, batch-to-batch moisture fluctuations will cause inconsistent color in the final product.
5. Application: Critical Decisions in Formulation & Manufacturing
For OEM factories and formulators, obtaining good raw materials is just the first step. GHK-Cu is as delicate as pure Vitamin C.
5.1 Dosing and pH System
- Golden Rule: Add during the cool-down phase (<40°C). Never include it in high-temperature emulsification.
- pH Window: Must be locked between 5.5 – 7.0. Too acidic causes copper ion dissociation and fading; too alkaline leads to copper hydroxide precipitation. Pre-buffering with Citric Acid/Arginine is recommended.
5.2 Incompatibility Guide
- Absolute Taboos: Disodium EDTA, Caprylhydroxamic Acid (CHA). These are powerful metal chelators that will instantly strip the copper ions.
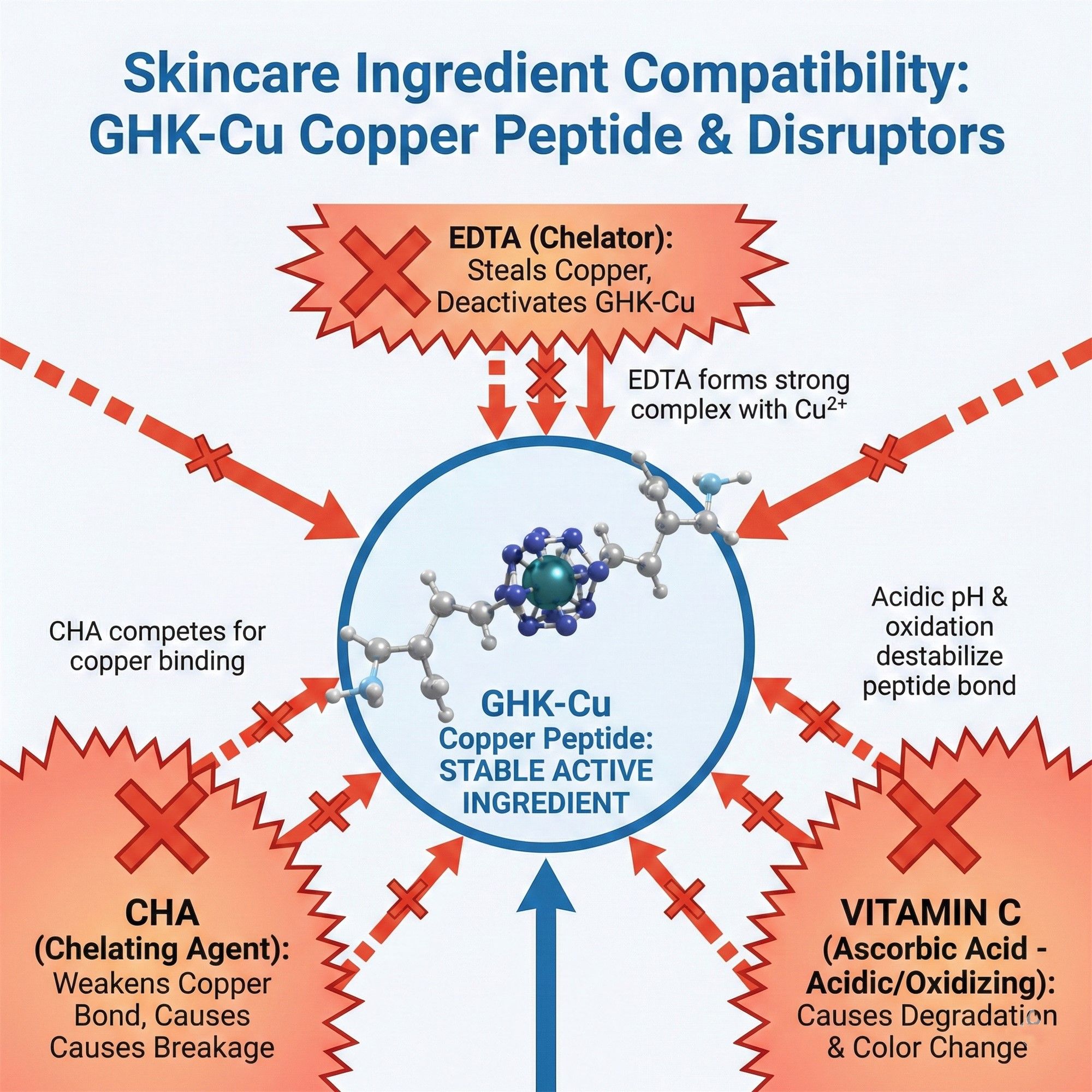
- Rheology Conflict: Avoid Carbomer (anionic); it causes ionic cross-linking leading to flocculation. Non-ionic thickeners like HEC or Xanthan Gum are recommended.
- Redox Reaction: Avoid direct combination with high concentrations of pure Vitamin C (Ascorbic Acid).
5.3 Dosage Forms
- Freeze-Dried Powder: Technically the optimal solution, completely solving shelf-life degradation issues.
- Encapsulation Technology: For GHK-Cu, the core value of encapsulation is not "penetration" (it has a low molecular weight and penetrates easily), but "Isolation"—creating a physical barrier when formulating with ingredients like Retinol.
6. Stability and Supply Chain Management
The stability of GHK-Cu is a systemic engineering challenge spanning from the warehouse to the shelf.
- Raw Material Storage: Combat thermodynamic entropy. Must be stored away from light, moisture, and at low temperatures (2-8°C). Once opened, it must be flushed with nitrogen and sealed; otherwise, it will rapidly absorb moisture and degrade.
- Finished Product Testing: Standard 45°C testing is insufficient. You must establish a pH Drift Curve (fluctuations >0.5 indicate danger) and monitor for microscopic precipitation.
- Packaging: Be wary of iron/zinc in pump springs causing metal displacement reactions.
7. Manufacturing Conclusions and Recommendations
Looking ahead to 2026, GHK-Cu remains the irreplaceable "Blue Blood Noble." For decision-makers, we offer three actionable recommendations:
- Procurement: Redefine the "Passing Line." Refuse raw materials that only provide purity based on area normalization. Require suppliers to provide NPC (Net Peptide Content) data and Elemental Analysis reports to verify the copper content follows the 1:1 logic.
- Process: Strictly adhere to pH and Temperature Red Lines. Adopt cold-process manufacturing, introduce buffer systems, and perform long-term pH drift monitoring.
- Verification: Introduce "Destructive" Testing. Add trace metal doping and high-intensity light tests during the R&D phase to expose formulation vulnerabilities early.
Conclusion
GHK-Cu is a mirror that reflects a brand's R&D depth. In the world of this blue molecule, high cost is not a choice, but a physical certainty.
When you decide not to compromise on low-price raw materials, what you deliver to consumers is no longer just a bottle of blue water, but an industrial promise regarding repair and renewal.
Scientific References:
Pickart, L. (2008). "The human tripeptide GHK and tissue remodeling". Journal of Biomaterials Science, Polymer Edition, 19(8), 969-988.
Maquart, F. X., Pickart, L., Laurent, M., Gillery, P., Monboisse, J. C., & Borel, J. P. (1988). "Stimulation of collagen synthesis in fibroblast cultures by the tripeptide-copper complex GHK-Cu". FEBS Letters, 238(2), 343-346.
Pickart, L., & Margolina, A. (2018). "Regenerative and Protective Actions of the GHK-Cu Peptide in the Light of the New Gene Data". International Journal of Molecular Sciences, 19(7), 1987.
Borkow, G. (2014). "Using Copper to Improve the Well-Being of the Skin". Current Chemical Biology, 8(2), 89-102.
Merrifield, R. B. (1963). "Solid Phase Peptide Synthesis. I. The Synthesis of a Tetrapeptide". Journal of the American Chemical Society, 85(14), 2149–2154.